New here?→ A0: What is CAR-T therapy? The big picture + index
New here?
→ A0: What is CAR-T therapy? The big picture + index
What you will learn
- The short answer: the appeal is “reset,” not chronic suppression
- 1) What “autoimmunity” means in plain language
- 2) Why B cells often matter (why CD19 shows up)
- 3) Suppression vs reset (the key mental model)
- 4) Where development is most visible (high-level buckets)
(Coming from solid tumors?)
→ A3: Why solid tumors are hard for CAR-T
→ B3: Solid-tumor CAR-T design toolkit
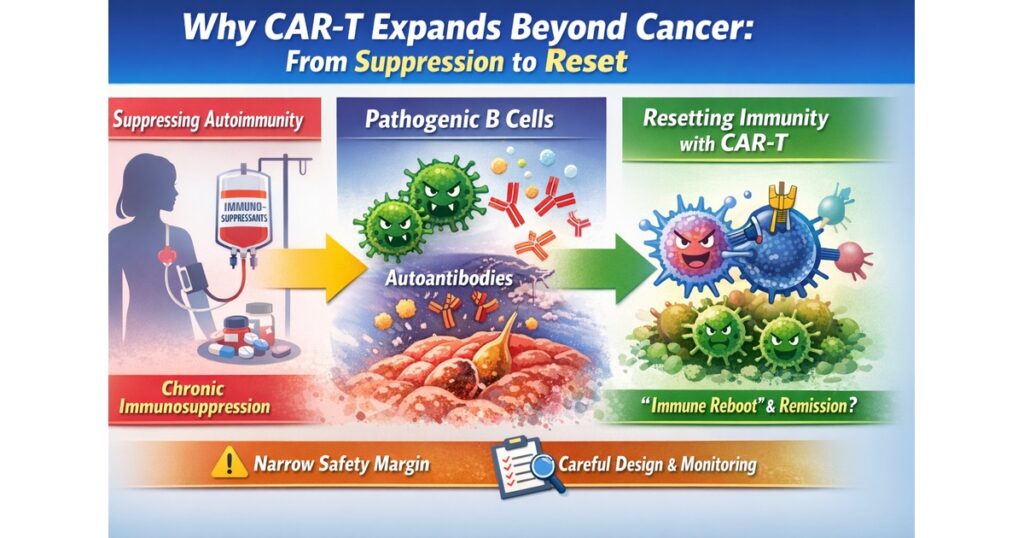
“From Suppression to Reset: Why CAR-T Moves Beyond Cancer”
The short answer: the appeal is “reset,” not chronic suppression
Many autoimmune diseases are driven by an immune system that mistakenly targets the body. Traditional care often relies on long-term immune suppression to keep inflammation down.
CAR-T enters this space because it offers a different conceptual goal: deeply depleting disease-driving immune components (often B cells) and enabling an “immune rebuild/reset.” Early clinical reports of CD19 CAR-T in refractory SLE triggered intense interest because they suggested durable remission with immune reconstitution features.
1) What “autoimmunity” means in plain language
Across diverse autoimmune conditions, a common pattern is:
- the immune system misidentifies self as a target
- inflammation persists and can damage organs (kidney, skin, joints, nerves, etc.)
- flares and relapses can occur
- long-term therapy often forces trade-offs (control vs infection risk)
This chronic/relapsing structure motivates the search for interventions that can change the immune set-point.
2) Why B cells often matter (why CD19 shows up)
In many autoimmune diseases, B cells can:
- support autoantibody production
- amplify inflammatory signaling
- shape other immune cell behaviors
This is why B-cell–directed therapies already exist in autoimmune care. CAR-T—especially CD19-directed CAR-T—can be viewed as a deeper, more transformative extension of this logic in selected settings.
3) Suppression vs reset (the key mental model)
Traditional suppression
- reduces inflammatory activity
- relapse risk may rise when therapy is tapered
- long-term immune suppression can increase infection risk
Reset concept
- aims to “rebuild” immune architecture after clearing key drivers
- in an ideal scenario, reduces the need for continuous immunosuppression
- but demands careful safety design, monitoring, and long-term follow-up
In refractory SLE, CD19 CAR-T reports described B-cell reappearance with a more naïve profile—supporting the “immune reconstitution” narrative.
4) Where development is most visible (high-level buckets)
4.1 Autoantibody/B-cell–driven arenas (e.g., SLE and lupus nephritis)
This is where the rationale is most direct and where clinical attention is concentrated. Industry development is active; for example, Kyverna’s KYV-101 is positioned for B-cell–driven autoimmune diseases including lupus nephritis, with trial activity documented publicly.
4.2 Organ-damaging systemic disease (e.g., systemic sclerosis)
When organ damage and long-term outcomes dominate, the value of durable control is high. Literature and reviews are expanding.
4.3 Neuro-immune conditions (e.g., selected neurologic autoimmune diseases)
B-cell involvement varies by disease, but the category is part of the broader “beyond cancer” conversation (details in B4).
5) The key difference from oncology: narrower toxicity tolerance
Compared with oncology, autoimmune applications generally operate under a tighter safety margin. As a result, design and operations tend to emphasize:
- lower immunogenicity constructs (often favoring “fully human” components)
- controllability and conservative escalation pathways
- long-term follow-up framing consistent with gene-modified cell therapy
6) “Does it cure autoimmunity in one shot?”
This is the most common misconception.
- Why people hope so: early refractory SLE reports were striking.
- What is still uncertain: cohort sizes are limited; optimal conditioning, dose, durability, and disease-by-disease fit remain under active evaluation. Reviews emphasize open questions.
7) Long-term follow-up (what it means for patients and families)
CAR-T sits within the gene-modified therapy landscape, where long-term follow-up (LTFU) is a core concept. FDA provides LTFU guidance for gene therapy products; autoimmune CAR-T programs must internalize that logic operationally.
Practically, families benefit most from:
- clear post-discharge call thresholds (fever, neuro change, infection signs)
- a simple plan for labs/visits and what each checks
- a shared understanding of why follow-up continues beyond “feeling better”
FAQ (search-aligned)
Q1. Is CAR-T already available for autoimmune diseases?
Mostly clinical trial settings, though high-impact reports and industry programs exist.
Q2. Why is CD19 so central?
Because it’s a practical B-cell marker and B cells are key drivers in many autoimmune patterns.
Q3. Is autoimmune CAR-T safer than cancer CAR-T?
Not a universal statement. The safety margin is tighter in autoimmunity, so design/monitoring is often more conservative and long-term follow-up is emphasized.
What’s next (B4)
B4 will organize autoimmune CAR-T by:
- disease bucket (SLE/LN, systemic sclerosis, neuro-immune, etc.)
- design philosophy (depletion/reset, reversibility, fully human, conditioning choices)
- representative programs/companies
Editors Note
This article covered the following key points:
- The short answer: the appeal is “reset,” not chronic suppression
- 1) What “autoimmunity” means in plain language
- 2) Why B cells often matter (why CD19 shows up)
- 3) Suppression vs reset (the key mental model)
Rather than fragmented news or definitions, the aim was to present these as a single connected flow. For one step deeper on the same theme, see the related articles below.
References
CD19 CAR-T in refractory SLE (Nature Medicine, 2022).
Autoimmune CAR-T review (Frontiers in Immunology, 2025).
KYV-101 trial information (Kyverna / ClinicalTrials.gov).
FDA LTFU guidance for gene therapy products (2020) + guidance hub.




Comments