Prefer the overview first?→ A4: Why CAR-T expands beyond cancer (autoimmunity as “reset”)
Prefer the overview first?
→ A4: Why CAR-T expands beyond cancer (autoimmunity as “reset”)
What you will learn
- Executive summary
- 1) The coordinate system: Target × Durability × Safety/Operations
- 2) Disease buckets: where programs look most “real”
- 3) Design philosophies: how autoimmune CAR differs from oncology CAR
- 4) Regulation and long-term safety: LTFU is built-in
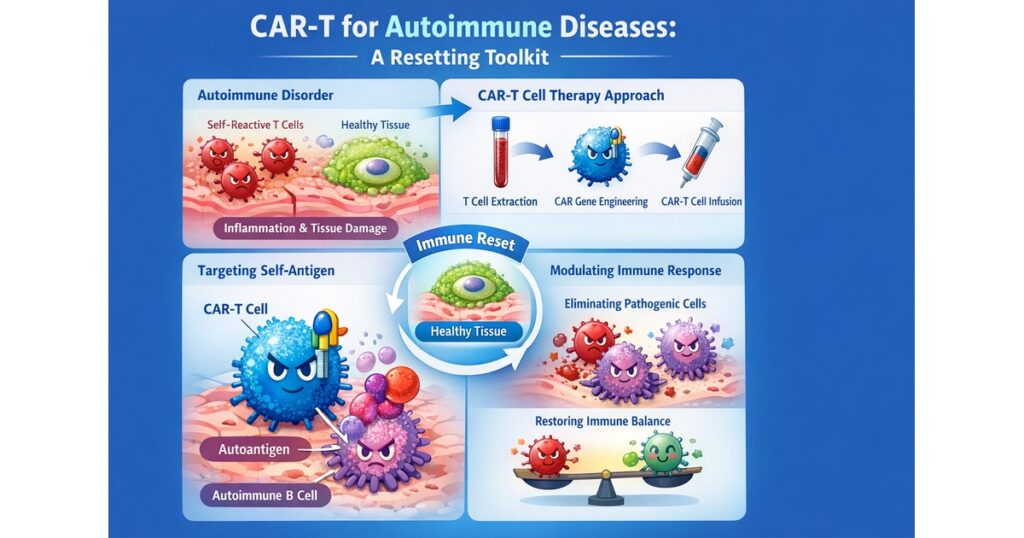
“Autoimmune CAR: Reset Toolkit (Target × Durability × Safety)”
Executive summary
Autoimmune CAR-T is shifting the goal from “killing tumor cells” to resetting immune circuitry and enabling durable remission. That changes both design priorities and operational constraints.
- The center of gravity is CD19 (B-cell targeting), reinforced by high-impact refractory SLE CD19 CAR-T reports and the subsequent wave of industry development.
- A practical industry frame includes Kyverna (KYV-101) and Cabaletta (CABA-201), both emphasizing fully human CD19 CAR-T concepts and severe autoimmune positioning.
- A distinct design path is BCMA-directed mRNA CAR-T (Cartesian’s Descartes-08), highlighting outpatient delivery, no preconditioning, and repeat dosing as a safety/operations strategy.
- Long-term follow-up (LTFU) is not optional; FDA’s LTFU guidance frames how sponsors plan for delayed risks in gene-modified therapies.
1) The coordinate system: Target × Durability × Safety/Operations
Target
- CD19: broad B-cell depletion logic (upstream driver in many autoantibody patterns)
- BCMA: plasma-cell/antibody-production axis (notably discussed in neurologic autoimmunity contexts)
Durability
- Integrating CAR-T: deep depletion → immune reconstitution/reset concept
- Transient mRNA CAR-T: controllability + outpatient repeat dosing (Descartes-08 narrative)
Safety/Operations
- conditioning intensity, monitoring pathway, and LTFU design are core value drivers
2) Disease buckets: where programs look most “real”
2.1 Autoantibody/B-cell–driven disease (SLE / lupus nephritis)
Refractory SLE CD19 CAR-T reports catalyzed the “immune reset” thesis and industry investment.
Kyverna positions KYV-101 (fully human anti-CD19 CAR-T) for B-cell–driven autoimmune disease including lupus nephritis, with trial information publicly documented.
2.2 Systemic/organ-damage disease (myositis / systemic sclerosis)
Cabaletta’s CABA-201 framing explicitly references severe autoimmune arenas including SLE, myositis, and systemic sclerosis.
2.3 Neuro-immune (myasthenia gravis and beyond)
Descartes-08 (BCMA-directed mRNA CAR-T) is described in the context of generalized MG, with an outpatient, no-preconditioning, repeat-infusion model.
3) Design philosophies: how autoimmune CAR differs from oncology CAR
Fully human binders (immunogenicity/re-treatment logic)
Both Kyverna and Cabaletta emphasize fully human CD19 CAR constructs as part of an autoimmune-appropriate design narrative.
Conditioning as a competitive design choice
Autoimmunity often runs on a tighter safety margin; “how much conditioning is needed” becomes a defining question. Descartes-08 is explicitly discussed as being delivered without preconditioning in key reports.
Transient mRNA CAR as an alternative safety/ops solution
mRNA CAR strategies emphasize controllability and operational scalability through repeat outpatient dosing.
4) Regulation and long-term safety: LTFU is built-in
FDA’s LTFU guidance provides a formal framework for designing long-term follow-up in gene therapy products, which is highly relevant for gene-modified cell therapies as well.
Operational meaning:
- follow-up is part of the therapy package
- durability claims require time, not headlines
- scaling autoimmune cell therapy implies scaling LTFU and post-treatment operations
5) What to watch in 12–24 months
- cross-disease reproducibility of CD19 CAR “reset” signals
- durability of drug-free remission with longer follow-up windows
- maturation of outpatient mRNA CAR delivery models
- conditioning optimization trade-offs (safety vs depth of reset)
Primary sources
Kyverna KYV-101 (LN trial / Fast Track).
Cabaletta CABA-201 (pipeline / RESET).
Cartesian Descartes-08 (MG; mRNA CAR).
FDA LTFU guidance (2020).
Editors Note
This article covered the following key points:
- Executive summary
- 1) The coordinate system: Target × Durability × Safety/Operations
- 2) Disease buckets: where programs look most “real”
- 3) Design philosophies: how autoimmune CAR differs from oncology CAR
Rather than fragmented news or definitions, the aim was to present these as a single connected flow. For one step deeper on the same theme, see the related articles below.




Comments