Prefer the beginner overview first?→ A0: What is CAR-T therapy? The big picture + index→ A4: Why CAR-T Is Expanding Beyond Cancer (Autoimmunity as an
Prefer the beginner overview first?
→ A0: What is CAR-T therapy? The big picture + index
→ A4: Why CAR-T Is Expanding Beyond Cancer (Autoimmunity as an “Immune Reset” Concept)
What you will learn
- The short answer: in vivo CAR aims to build CAR cells inside the body
- 1) Why in vivo CAR is being pursued: removing classic CAR-T bottlenecks
- 2) Three high-level implementation patterns
- 3) Where the field is now: academic validation + clinical momentum
- 4) The upside (what could be meaningfully better)
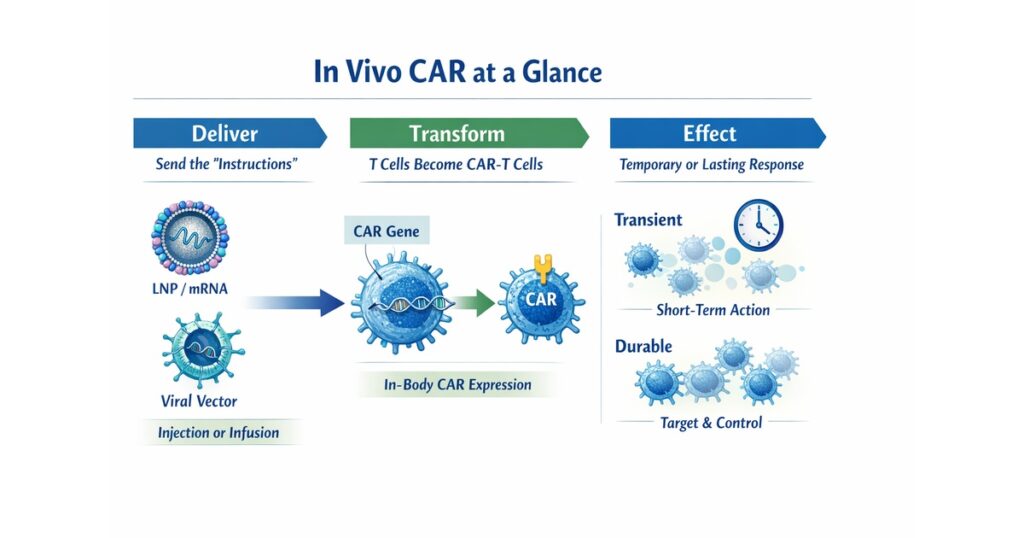
“In vivo CAR at a glance: dose → in-body CAR expression → transient vs durable effects”
The short answer: in vivo CAR aims to build CAR cells inside the body
Conventional CAR-T is made outside the body (collect cells → engineer/expand → infuse).
In vivo CAR aims to deliver the “instructions” so that CAR cells are generated inside the patient.
Think:
- Conventional CAR-T: manufacture outside → ship back in
- In vivo CAR: deliver instructions → manufacture at the point of care (in vivo)
The motivation is to remove bottlenecks in time, infrastructure, and access.
1) Why in vivo CAR is being pursued: removing classic CAR-T bottlenecks
Ex vivo CAR-T often implies:
- manufacturing turnaround time and logistics
- specialized sites and operational complexity
- high cost and limited scalability
- patient-by-patient manufacturing constraints
In vivo CAR tries to move closer to an “off-the-shelf dosing” model.
2) Three high-level implementation patterns
Pattern A: deliver mRNA (e.g., LNP) → transient CAR expression
mRNA delivery can yield time-limited CAR expression, which can be attractive for safety and controllability.
Pattern B: deliver DNA (e.g., viral vectors) → potentially longer expression
Durability may improve, but long-term safety and follow-up become more central.
Pattern C: in vivo gene editing/integration (future-facing)
High specificity is required; could become “true in vivo cell therapy” if solved.
3) Where the field is now: academic validation + clinical momentum
Academic proof-of-concept (Science, 2025)
A Science paper reported in vivo CAR-T generation via CD8-targeted LNP delivering anti-CD19 CAR mRNA.
Clinical momentum (Capstan, 2025)
Capstan announced initiation of a Phase 1 trial for CPTX2309, an in vivo anti-CD19 CAR-T using targeted LNPs, emphasizing tunable/dose-dependent CAR expression for B-cell–mediated autoimmune disorders.
4) The upside (what could be meaningfully better)
- faster access (less manufacturing delay)
- broader reach (less site dependence)
- repeatability (especially for mRNA approaches)
- better cost structure vs individualized manufacturing (in principle)
5) The key risks (what must be solved)
Risk 1: delivery specificity (off-target transfection)
Targeted delivery is the core problem—hence CD8-targeted LNP concepts.
Risk 2: controllability (how to avoid runaway activity)
Dose, interval, and variability across patients must be engineered into protocols; transient expression helps but is not a full solution.
Risk 3: long-term safety and follow-up
Gene-modified therapy logic brings long-term follow-up expectations; FDA LTFU guidance frames this landscape.
6) A simple way to read in vivo CAR news (4 checks)
- What is delivered? (mRNA/LNP vs DNA/vector)
- Which cells are targeted?
- Is expression transient or durable?
- What is the safety/control and follow-up design?
Next (B5)
B5 will cover implementation details: delivery engineering, dose-control logic, safety mechanisms, and regulatory/CMC implications.
Editors Note
This article covered the following key points:
- The short answer: in vivo CAR aims to build CAR cells inside the body
- 1) Why in vivo CAR is being pursued: removing classic CAR-T bottlenecks
- 2) Three high-level implementation patterns
- 3) Where the field is now: academic validation + clinical momentum
Rather than fragmented news or definitions, the aim was to present these as a single connected flow. For one step deeper on the same theme, see the related articles below.
References
Science (2025): CD8-targeted LNP in vivo CAR-T generation.
Capstan (2025): CPTX2309 Phase 1 initiation (tLNP; tunable CAR expression).
Trends in Biotechnology (2025): in vivo CAR technology review.
FDA (2020): Long-term follow-up guidance for gene therapy products.




Comments