If you want the beginner version first:→ A5: What is in vivo CAR?
If you want the beginner version first:
→ A5: What is in vivo CAR? “Making CAR cells inside the body” explained simply
What you will learn
- Executive summary
- 1) Delivery: the “core” of in vivo CAR
- 2) Control: dosing design becomes the product
- 3) Safety: off-target is the main battlefield in vivo
- 4) CMC/Reg: in vivo CAR wins or loses on drug-like reproducibility
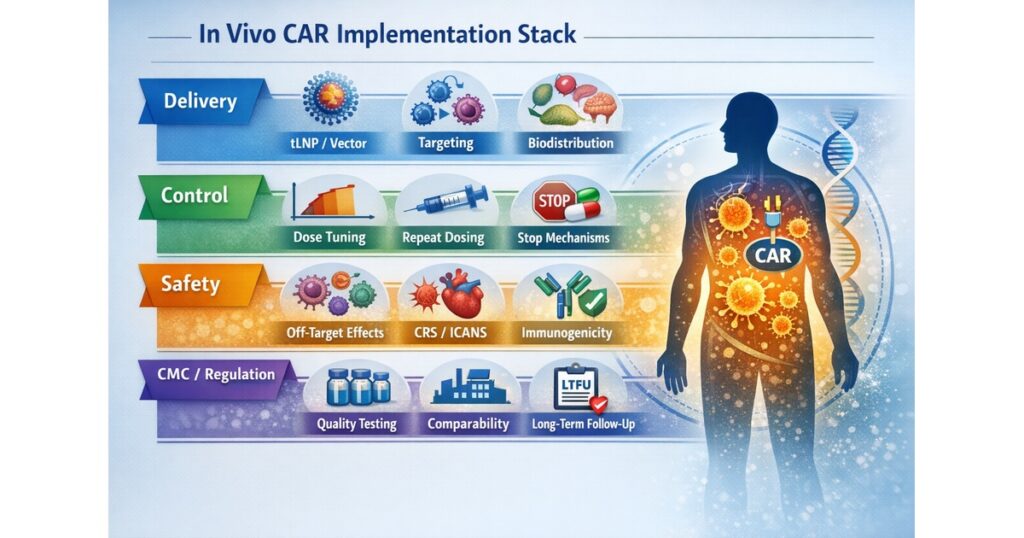
“In Vivo CAR Implementation Stack”
- Delivery (tLNP/vector, targeting, biodistribution)
- Control (dose-dependent, repeat dosing, stop mechanisms)
- Safety (off-target, CRS/ICANS, immunogenicity, long-term risk)
- CMC/Reg (specs, release tests, comparability, LTFU)
Executive summary
The essence of in vivo CAR is not simply “moving CAR-T manufacturing into the body.” It is making in-body biodistribution, control, and safety reproducible as a drug. The decisive bottlenecks converge on four points:
- Where and how much reaches the intended cells (targeted biodistribution)
- How strong and how long activity is allowed (tunable controllability)
- What happens in unintended cells/tissues (off-target toxicology)
- Whether quality can be standardized and scaled (CMC, release tests, comparability)
Two concrete catalysts accelerated this field: the Science (2025) report using CD8-targeted tLNP to deliver CAR mRNA into T cells in vivo, and Capstan’s announcement of a Phase 1 start for CPTX2309.
(science.org)
In addition, industry confidence moved another step when AbbVie’s large acquisition of Capstan’s in vivo CAR platform was reported.
(nature.com)
1) Delivery: the “core” of in vivo CAR
1-1) What is delivered: mRNA (transient) vs DNA/vector (longer)
- mRNA (LNP/tLNP): expression turns off over time—this can function as a safety valve. It also enables repeat dosing and dose tuning.
(science.org) - DNA/vector: may enable longer expression, but long-term safety and long-term follow-up requirements typically become heavier (and whether that is acceptable in autoimmunity becomes a central question).
The most “clinically close” current trend is tLNP × mRNA × targeting to secure reversibility and controllability.
(science.org)
1-2) Which cells receive it: CD8 targeting as a signature design philosophy
Science (2025) proposes an implementation where tLNP targeting to specific T-cell subsets is used to generate CAR-T in vivo. The key is pushing back against the classic “IV → liver trap” problem via targeting.
(science.org)
KPIs readers should look for
- Transfection efficiency in the intended target cells
- Off-target expression in non-target cells (e.g., liver, monocytes, endothelium, etc.)
- Biodistribution not only in blood but also in tissues (lymph nodes, spleen, inflamed sites)
1-3) Biodistribution defines value: autoimmunity vs cancer
- Autoimmunity: if the goal is to reset B-cell/autoantibody drivers, designs that reach lymphoid compartments and inflamed sites create value.
- Cancer: reaching the tumor site defines value (this connects directly to the “access” barrier in B3).
Capstan’s CPTX2309 description emphasizes in vivo engineering of CD8+ CAR T cells, B-cell depletion across blood and tissues, and a possible shift toward more “naïve-like” B-cell reconstitution in preclinical work.
(forbion.com)
2) Control: dosing design becomes the product
2-1) How to establish dose-dependence (tunable control)
Capstan highlights “tunable and dose-dependent levels of CAR expression.” This is especially important in autoimmunity, where the safety margin is tighter and over-activation must be avoided.
(forbion.com)
The key question is how stable “dose → expression → efficacy/toxicity” remains under:
- inter-individual variability (body size, immune state)
- inflammatory state (vascular permeability, target-cell abundance)
2-2) Conditions required for repeat dosing
mRNA/tLNP systems are attractive for repeat dosing, but repeat dosing only works if these constraints are addressed:
- Immunogenicity to LNP components or binding moieties (neutralization risk)
- interval optimization (balancing efficacy and safety)
- unexpected inflammation accumulating across repeats
This area often becomes “operationalized” during clinical development rather than fully pre-solved in papers.
2-3) How “stopping” is ensured
In ex vivo CAR-T, “suicide switches” and drug-inducible shutdown are often discussed. In in vivo CAR, the most powerful stop mechanisms can be:
- expression naturally ends (mRNA)
- stop dosing
Conversely, for long-expression approaches, the design requirements for robust stop mechanisms rise sharply.
3) Safety: off-target is the main battlefield in vivo
3-1) On-target effects (e.g., B-cell depletion) are not “side effects” but design intent
For CD19 targeting, B-cell reduction is intended. The operational challenge is what follows:
- infection risk (immunity down)
- quality of immune reconstitution (how cells return)
- vaccination and prophylaxis planning
CPTX2309 highlights deep B-cell depletion and “naïve-leaning” reconstitution as preclinical signals; clinical reproducibility is the next focal point.
(forbion.com)
3-2) Off-target expression: the highest-priority risk
If hepatocytes, monocytes, or other unintended cells express CAR, unexpected toxicity can occur. Therefore, the package must be evaluated as a set:
- targeted delivery (e.g., CD8 targeting)
(science.org) - localized/minimized expression where needed
- duration control (mRNA)
3-3) CRS/ICANS should be assumed “not zero”
If immune activation becomes strong, CRS/ICANS-like events can occur in principle. Thus, the toxicity management philosophy from B2 (early detection and stepwise escalation) remains relevant—although frequency and patterns may differ.
3-4) Long-term safety: LTFU logic from gene therapy
FDA provides a long-term follow-up (LTFU) guidance framework for gene therapy products. As in vivo CAR approaches align more with “gene therapy/gene modification” persistence, follow-up design becomes heavier and more central.
(fda.gov)
This is part of why transient mRNA designs can be easier to accept in autoimmunity.
4) CMC/Reg: in vivo CAR wins or loses on drug-like reproducibility
Ex vivo CAR-T historically centers on cell-product release testing. In vivo CAR shifts quality focus to:
- LNP properties (particle size, encapsulation, purity)
- binding function of antibody/ligand moieties
- mRNA quality (sequence, modifications, cap, dsRNA impurities)
- potency definition: how to standardize “what % of T cells are CAR-programmed and functional”
The hardest problem is potency.
Even at the same dose, T-cell state differs across patients, changing programming, expansion, and effect. The winning approach is being able to separate “product quality” from “patient biology” while maintaining a reproducible clinical profile.
5) Industry dynamics: what AbbVie’s large acquisition signals
Nature reported AbbVie’s ~$2.1B acquisition of Capstan and framed it as a platform bet on in vivo CAR technology (mRNA-LNP reprogramming of immune cells).
(nature.com)
What readers should take from this
- A modality “path to winning” is becoming visible (especially for autoimmunity)
- Competition shifts from “science possibility” to “implementation (delivery × control × CMC)”
6) Pro evaluation checklist for in vivo CAR news (copy/paste)
- What is delivered? (mRNA vs DNA)
- How is targeting achieved? (e.g., CD8 targeting)
(science.org) - How comprehensively is off-target evaluated? (liver, monocytes, endothelium, bone marrow, etc.)
- Evidence that dose-dependent control truly holds
(forbion.com) - Repeat dosing feasibility (immunogenicity/neutralization)?
- How is potency defined and standardized (release strategy)?
- How is LTFU/long-term follow-up designed?
(fda.gov)
Next (A6/B6 flow)
Per the plan, the most natural next step is:
A6 (Beginner): Manufacturing & access—why is CAR-T expensive, and what can change?
→ B6 (Expert): COGS, supply chain, site requirements, regulation and commercialization
Primary sources
- Science (2025): CD8-targeted tLNP strategy for in vivo CAR-T generation
(science.org) - Capstan (2025): CPTX2309 Phase 1 initiation (dose-dependent CAR expression, etc.)
(forbion.com) - FDA (2020): Gene therapy LTFU guidance
(fda.gov) - Nature (2025): AbbVie acquisition of Capstan (reported)
(nature.com)
Editors Note
This article covered the following key points:
- Executive summary
- 1) Delivery: the “core” of in vivo CAR
- 2) Control: dosing design becomes the product
- 3) Safety: off-target is the main battlefield in vivo
Rather than fragmented news or definitions, the aim was to present these as a single connected flow. For one step deeper on the same theme, see the related articles below.



Comments