How to use this series (two-layer linkage: Beginner → Expert)
This series explains CAR-T therapy in a stepwise way so that readers who are new to the term can first grasp the big picture (Layer A), and then move to the expert layer (Layer B) for “companies, targets (e.g., CD19/BCMA), CAR design, clinical evidence, and regulatory/CMC considerations.”
- Layer A (Beginner-friendly): high-level categories and the overall picture, minimal jargon with plain explanations.
- Layer B (Expert): key disease names, company/program landscape, CAR engineering, evidence, and FDA/regulatory logic.
- Posting cadence: A → B → A → B… (daily, alternating).
Medical disclaimer: This article is for general education only and is not medical advice. Treatment decisions must be made with your care team.
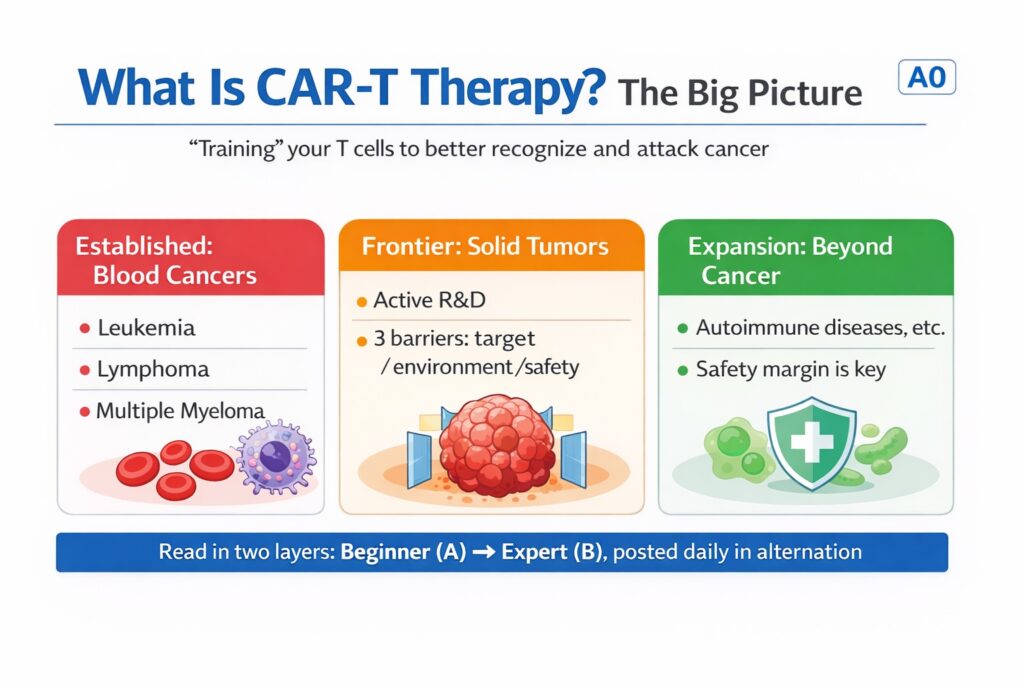
Figure: CAR-T at a glance — strongest track record in blood cancers; active R&D in solid tumors and expanding into non-cancer diseases. This series links a beginner layer (A) to an expert layer (B).
What is CAR-T therapy? (the short answer)
CAR-T therapy is a treatment in which a patient’s own T cells (immune cells) are collected, modified outside the body to recognize cancer cells more effectively, and then infused back into the patient. In plain terms, it is a way of “training your immune cells to better find and attack cancer.”
CAR-T is most established in blood cancers (such as certain leukemias, lymphomas, and multiple myeloma), where multiple products have been approved by the U.S. FDA. At the same time, the field is pushing into solid tumors and even non-cancer diseases (notably autoimmune conditions).
Do I need to memorize the acronym “CAR”? Not really.
CAR stands for Chimeric Antigen Receptor. You don’t need to memorize the term. Just think of CAR as a sensor (an “antenna”) placed on T cells so they can better recognize a specific target on diseased cells.
- T cell: one of the immune system’s key “fighter” cells.
- CAR: a recognition sensor that helps T cells detect a target (an “antigen”).
The real-world treatment journey (what patients and families want to know)
CAR-T is not a simple one-time infusion with no preparation. The process usually includes:
- Cell collection: T cells are collected from blood (often similar to apheresis).
- Manufacturing: the cells are modified to express the CAR (this takes time).
- Pre-treatment (often): short therapy to help CAR-T cells expand and work effectively.
- Infusion: CAR-T cells are given back to the patient.
- Monitoring: careful observation for side effects and response.
Monitoring matters because CAR-T can trigger strong immune activation, and early detection/management of side effects is a key part of safe care.
Where CAR-T is strongest today: blood cancers (big categories)
CAR-T has the clearest clinical impact today in blood cancers. For beginner readers, it helps to start with these broad categories (specific disease names appear in the expert layer):
- Leukemia: cancers involving blood-forming cells (multiple subtypes exist).
- Lymphoma: cancers of immune cells (many subtypes; CAR-T is important in several).
- Multiple myeloma: a blood cancer of plasma cells; CAR-T targets (like BCMA) are central here.
Why solid tumors are harder (just the essentials)
Solid tumors (e.g., lung, stomach, colon cancers) are a major frontier, but they are harder than blood cancers because:
- Targets can be heterogeneous: not every tumor cell displays the same “marker.”
- The tumor microenvironment suppresses immunity: immune cells can be blocked or exhausted.
- Safety design is tougher: some targets may also exist on healthy tissues.
Later posts will explain the engineering strategies that try to overcome these barriers (e.g., multi-target designs, logic gates, armored CARs, local delivery).
Expansion beyond cancer (autoimmune diseases and more)
CAR-T concepts are increasingly explored in autoimmune diseases, where the immune system mistakenly attacks the body. Traditional care often relies on long-term immune suppression. CAR-based approaches may offer a different concept: resetting or rebalancing immune drivers of disease.
However, autoimmune applications demand a wider safety margin than oncology. Therefore, researchers are exploring strategies aimed at safer control (including “reversible” designs). The expert layer will cover the leading approaches and programs.
Side effects (without fear, but with clarity)
The two most discussed categories of CAR-T side effects are:
- Strong inflammatory reactions (often described clinically as CRS).
- Temporary neurologic symptoms (often discussed as ICANS and related terms).
Clinical teams have built standardized monitoring and management frameworks. Long-term safety monitoring is also an important part of the field’s evolution, and FDA communications reflect that reality (explained in the expert layer).
How to read CAR-T news (3 quick checks)
- Which arena? Blood cancer vs. solid tumor vs. non-cancer.
- Which stage? Lab research vs. clinical trial vs. FDA approval vs. routine practice.
- Who was treated? Disease subtype, prior therapies, disease burden, fitness, etc.
Series index (daily A/B alternation)
Replace “Coming soon” with live links as you publish.
- A0 (this post) [Beginner]: What is CAR-T? The big picture + series index
- A1 [Beginner]: Where CAR-T is used today (high-level categories) — (Coming soon)
- B1 [Expert]: Approved CAR-T landscape (products, targets, companies, design, key disease names) — (Coming soon)
- A2 [Beginner]: Side effects without panic (what to expect, why monitoring matters) — (Coming soon)
- B2 [Expert]: Toxicity management, site operations, long-term safety, regulatory logic — (Coming soon)
- A3 [Beginner]: Why solid tumors are hard (the big picture) — (Coming soon)
- B3 [Expert]: Next-gen CAR engineering strategies (armored, multi-target, logic-gated, local delivery) — (Coming soon)
- A4 [Beginner]: Why CAR approaches are expanding beyond cancer — (Coming soon)
- B4 [Expert]: Autoimmune CAR programs and design philosophies — (Coming soon)
- A5 [Beginner]: “In vivo CAR” — what it means and why it matters — (Coming soon)
- B5 [Expert]: In vivo CAR: delivery, control, safety, regulatory implications — (Coming soon)
Mini glossary (beginner-friendly)
- Immune system: the body’s defense network that detects and removes threats and abnormal cells.
- T cell: a key immune cell type that can kill abnormal or infected cells.
- Antigen / target: a “marker” the immune system (or CAR) can recognize.
- Blood cancers: cancers affecting blood/immune cells (e.g., leukemia, lymphoma, myeloma).
Primary references (FDA)
- FDA: Approved Cellular and Gene Therapy Products
https://www.fda.gov/vaccines-blood-biologics/cellular-gene-therapy-products/approved-cellular-and-gene-therapy-products - FDA: REMS elimination for autologous CAR-T
https://www.fda.gov/news-events/press-announcements/fda-eliminates-risk-evaluation-and-mitigation-strategies-rems-autologous-chimeric-antigen-receptor - FDA: Boxed warning on T-cell malignancies following certain CAR-T therapies
https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/fda-requires-boxed-warning-t-cell-malignancies-following-treatment-bcma-directed-or-cd19-directed - FDA: CAR-T product development guidance (CMC, etc.)
https://www.fda.gov/regulatory-information/search-fda-guidance-documents/considerations-development-chimeric-antigen-receptor-car-t-cell-products
Comments