Key Takeaways
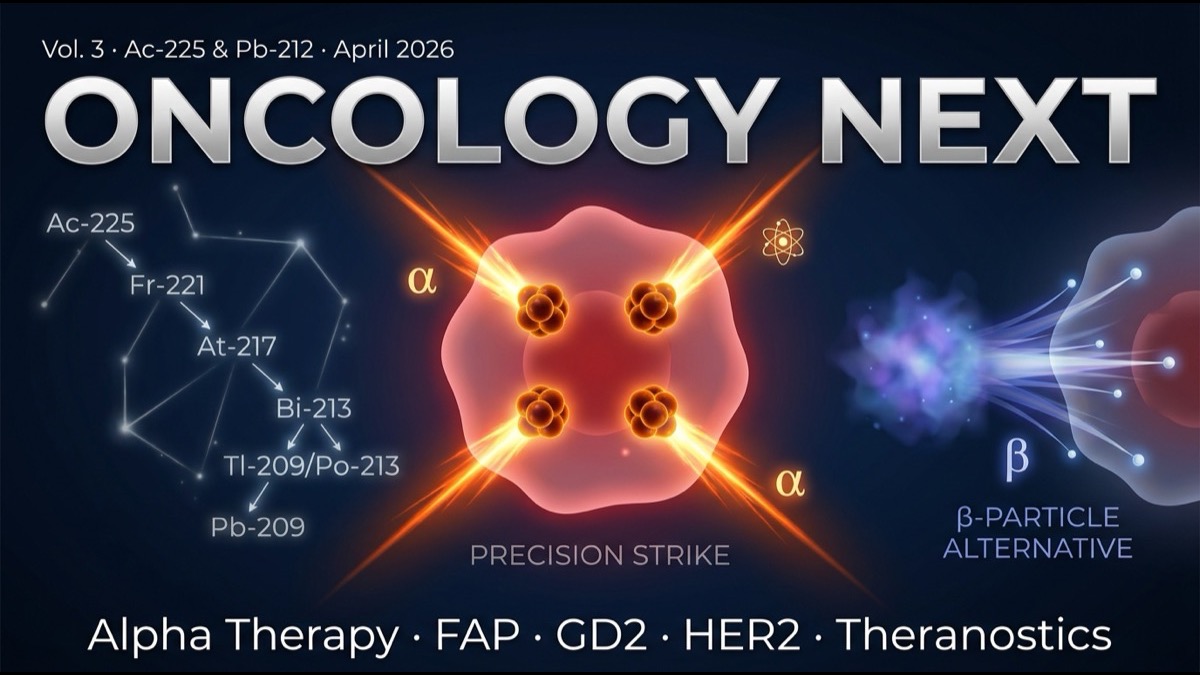
- Nuclear medicine’s next decade will see the “β-emitter (¹⁷⁷Lu) to α-emitter (²²⁵Ac, ²¹²Pb)” shift accelerate. α-emitters are ~100× more cytotoxic than β with shorter range (a few cell diameters), enabling pinpoint tumor cell attacks while limiting normal tissue impact.
- Major α therapy pipelines: ²²⁵Ac-PSMA (prostate, Pluvicto α), ²²⁵Ac-FAP (generalist solid tumors), ²²⁵Ac-DOTATATE (NET, Lutathera α — BMS RYZ101), ²¹²Pb-DOTAMTATE (NET, Perspective Therapeutics). Multiple in Phase 1-2.
- New target expansion: FAP (fibroblast activation protein, generalist solid tumors), GD2 (neuroblastoma, glioblastoma), HER2 (breast, gastric), αvβ6 integrin (pancreatic, head/neck) — beyond historic limited targets.
- The manufacturing wall: ²²⁵Ac global production capacity remains limited. Oak Ridge National Lab (US), Karlsruhe (Germany), PSI (Switzerland), JAEA (Japan) — limited supply sources. Commercial-scale expansion is the largest 3-5 year challenge.
Introduction — “Beyond the β Era”
Volumes 1 and 2 covered the current nuclear medicine market centered on ¹⁷⁷Lu (β-emitter). Pluvicto and Lutathera both use ¹⁷⁷Lu; over 80% of the field is β therapy.
But radiobiologically, α-emitters have far more therapeutic suitability than β. This article explores α therapy science, clinical pipelines, manufacturing constraints, and the next-decade direction for nuclear medicine.
Main Body
1. α vs β — Radiobiology Fundamentals
| Property | α | β |
|---|---|---|
| Particle | Helium-4 nucleus | Electron |
| Energy | 5-9 MeV | 0.5-2 MeV |
| Range (in tissue) | 40-100 µm (a few cell diameters) | 1-10 mm |
| LET (linear energy transfer) | High (80-100 keV/µm) | Low (0.2 keV/µm) |
| Cytotoxicity | ~100× higher | Standard |
| DNA double-strand breaks | Frequent (hard to repair) | Mainly single-strand (repairable) |
| Normal tissue impact | Limited (short range) | Broad (includes repairable tissues) |
The α property of “short range + high LET” is ideal for tumor therapy. Within target cells, α causes many DNA double-strand breaks while limiting impact on adjacent normal tissue. This produces a “micro-precision-guided munition”-like effect.
2. ²²⁵Ac Clinical Pipeline
²²⁵Ac-PSMA-617 (Pluvicto α version):
- ²²⁵Ac has 9.9-day half-life with 4-step α-decay cascade releasing 4 α particles.
- South African Sahmdz trial (2017-) showed responses in Pluvicto (¹⁷⁷Lu)-failed patients.
- Novartis internal development, Bayer strategic partnership for multicenter trials.
²²⁵Ac-DOTATATE (RYZ101, BMS × RayzeBio):
- NET treatment, evaluated in Lutathera-failed patients.
- Phase 2 ongoing, early response data favorable.
²²⁵Ac-FAP-2286 (Aktis Oncology / Lilly × Mariana ecosystem):
- FAP is broadly expressed on tumor-associated fibroblasts.
- Solid tumors broadly (pancreatic, colorectal, breast, lung, head/neck).
- Phase 1 progressing.
3. ²¹²Pb Clinical Pipeline
²¹²Pb characteristics: 10.6-hour half-life is short but α-cascade decay scheme. Therapeutic effect develops in shorter time than ²²⁵Ac, advantageous for shorter patient hospital stay.
Major pipelines:
- ²¹²Pb-DOTAMTATE (Perspective Therapeutics): NET treatment, Phase 2.
- ²¹²Pb-PSMA: prostate cancer, Phase 1.
- ²¹²Pb-FAP: generalist solid tumors, preclinical → Phase 1.
4. New Target Expansion
Direction beyond PSMA/SSTR limited targets:
- FAP: generalist solid tumors (>70% expressing), Aktis / Lilly × Mariana / Fusion competing.
- GD2: neuroblastoma (pediatric), glioblastoma, SCLC — fusion of immunotherapy (dinutuximab, GD2 CAR-T) and nuclear medicine.
- HER2: breast and gastric cancer adding α therapy on top of targeted drugs (Trastuzumab, T-DXd).
- αvβ6 integrin: pancreatic, head/neck, lung cancers high expression.
- CAIX (Carbonic Anhydrase IX): renal cell carcinoma (Telix TLX250).
- NTSR1 (Neurotensin Receptor 1): pancreatic, colorectal, prostate (Fusion FPI-2059).
- uPAR (Urokinase Plasminogen Activator Receptor): breast, colorectal cancer (Curasight).
5. Manufacturing Wall — ²²⁵Ac Global Supply
The largest wall for clinical α therapy is production capacity. Major ²²⁵Ac production facilities:
- Oak Ridge National Laboratory (US, Tennessee): ²²⁹Th separation, hundreds-thousands mCi/year.
- Karlsruhe Institute (Germany): major European supplier.
- Paul Scherrer Institute (PSI, Switzerland): neutron capture (²²⁶Ra → ²²⁷Ac → ²²⁵Ac).
- JAEA (Japan): QST partnership, building ²²⁵Ac production capability.
- Commercial ²²⁵Ac production: Novartis, BMS, NorthStar Medical Radioisotopes investing in expansion.
2025 global ²²⁵Ac supply is approximately 2-5 Ci/year — sufficient for Phase 1-2 trials but commercial scale (tens of Ci/year) expansion is essential.
6. Theranostics — “Diagnosis and Therapy with the Same Molecule”
A unique evolution in nuclear medicine is theranostics (therapeutics + diagnostics). Same target molecule labeled with γ / positron emitters for diagnosis and α / β emitters for therapy.
Examples:
- Diagnosis: ⁶⁸Ga-PSMA-11 (PET); Treatment: ¹⁷⁷Lu-PSMA-617 (β) or ²²⁵Ac-PSMA-617 (α).
- Diagnosis: ⁶⁸Ga-DOTATATE; Treatment: ¹⁷⁷Lu-DOTATATE or ²²⁵Ac-DOTATATE.
- Diagnosis: ⁶⁴Cu-FAPI; Treatment: ⁶⁷Cu-FAPI (Clarity Pharmaceuticals’ Cu-64/Cu-67 pair).
Theranostics is the ideal of “individually optimized treatment” — pre-treatment PET diagnosis confirms target expression, then treatment is administered only to high-expression patients, maximizing efficacy and safety.
7. AI Integration with Nuclear Medicine
Areas of AI integration in the next decade:
- Individual dosimetry optimization: AI-driven dose decisions based on patient size, renal function, tumor burden.
- Target expression prediction: PET image + AI for treatment response prediction.
- Manufacturing management: real-time optimization of radioisotope production and distribution.
- Clinical trial design: optimization of multi-isotope, multi-target combinations.
8. Limitations and Caveats
First, long-term safety of α therapy. ²²⁵Ac 4-step α decay is theoretically powerful but secondary malignancy risk requires long-term evaluation.
Second, manufacturing bottleneck. Commercial-scale expansion of ²²⁵Ac global supply is a 3-5 year challenge.
Third, cost. α therapy production cost is higher than ¹⁷⁷Lu, potentially raising per-patient cost.
Fourth, facility requirements. α therapy may require even stricter facility requirements than β.
Summary
- Nuclear medicine’s next decade: “β (¹⁷⁷Lu) → α (²²⁵Ac, ²¹²Pb)” shift. α has 100× cytotoxicity, short range protects normal tissue.
- Major α pipelines: ²²⁵Ac-PSMA, ²²⁵Ac-FAP, ²²⁵Ac-DOTATATE (RYZ101), ²¹²Pb-DOTAMTATE.
- New target expansion: FAP, GD2, HER2, αvβ6, CAIX, NTSR1, uPAR — generalist solid tumor application.
- Manufacturing wall: commercial-scale ²²⁵Ac expansion is 3-5 year challenge. Oak Ridge, Karlsruhe, PSI, JAEA major facilities.
- Theranostics: diagnosis and treatment with same target molecule. Ideal of individualized treatment.
- AI integration: dosimetry optimization, target expression prediction, manufacturing management, clinical trial design.
Series Synthesis
Across the three-volume “Nuclear Medicine Frontier” series, we dissected the structural changes in nuclear medicine starting from BioSpace’s April 2026 industry analysis. Volume 1: Pluvicto / Lutathera success and Vertical Integration. Volume 2: competitive players Lantheus, Bayer, Telix, Lilly × Mariana, BMS × RayzeBio, AZ × Fusion. Volume 3 (this article): α-emitter era and next-generation pipelines. Nuclear medicine is steadily evolving from “niche specialty” to “one of the major axes of cancer therapy in the 2030s.”
My Thoughts and Outlook
The deepest structural insight from this series is that “nuclear medicine is not a single drug but an ecosystem competition.” Isotope production networks, GMP synthesis, distribution, specialized facilities, specialized personnel — the vertically integrated company that can control all of this is establishing dominance in this industry structure.
Three structural implications for the global ecosystem. First, isotope production becomes strategic infrastructure for “medical sovereignty.” Countries are recognizing that domestic production of ²²⁵Ac, ¹⁷⁷Lu, ²¹²Pb is as important as semiconductor capability — the EU is investing heavily in EURADOS / SCK-CEN Belgium expansion, the US Department of Energy is funding Oak Ridge production, and emerging Asian centers (South Korea KIRAMS, India BARC) are scaling up. Second, the boundary between “drug discovery” and “physics-engineering” is dissolving. Nuclear medicine increasingly resembles semiconductor manufacturing in requiring vertically integrated, time-critical, highly engineered production. The 2030s will see “isotope-pharmaceutical” become a major industry category, distinct from “small-molecule” or “biologics.” Third, theranostics personalization becomes the gold standard. Pre-treatment PET diagnosis + AI prediction + individualized α / β therapy combination will replace one-size-fits-all dosing. The first FDA-approved AI-driven theranostic optimization platform may emerge by 2027-28.
2026 is the year AI is rapidly commoditizing knowledge work. Nuclear medicine sits squarely in the “AI cannot replace” zone, and AI augments it powerfully — physical isotope production, clinical decision-making, regulatory navigation cannot be replaced by AI, but AI accelerates dosimetry, image analysis, and trial design. The next decade will see nuclear medicine evolve from specialty market into a major axis of cancer treatment alongside chemotherapy, radiation, targeted therapy, immunotherapy, and cell therapy.
Edited by the Morningglorysciences team.


Comments