Key Takeaways
- The April 2026 issue of Nature Medicine published a landmark paper validating the real-world clinical utility of tumor whole-genome sequencing (WGS) in solid cancers across multicenter, large-scale real-world cohorts. The paper provides decisive evidence on a long-debated question — whether WGS, scientifically rich but economically uncertain, has implementable clinical value.
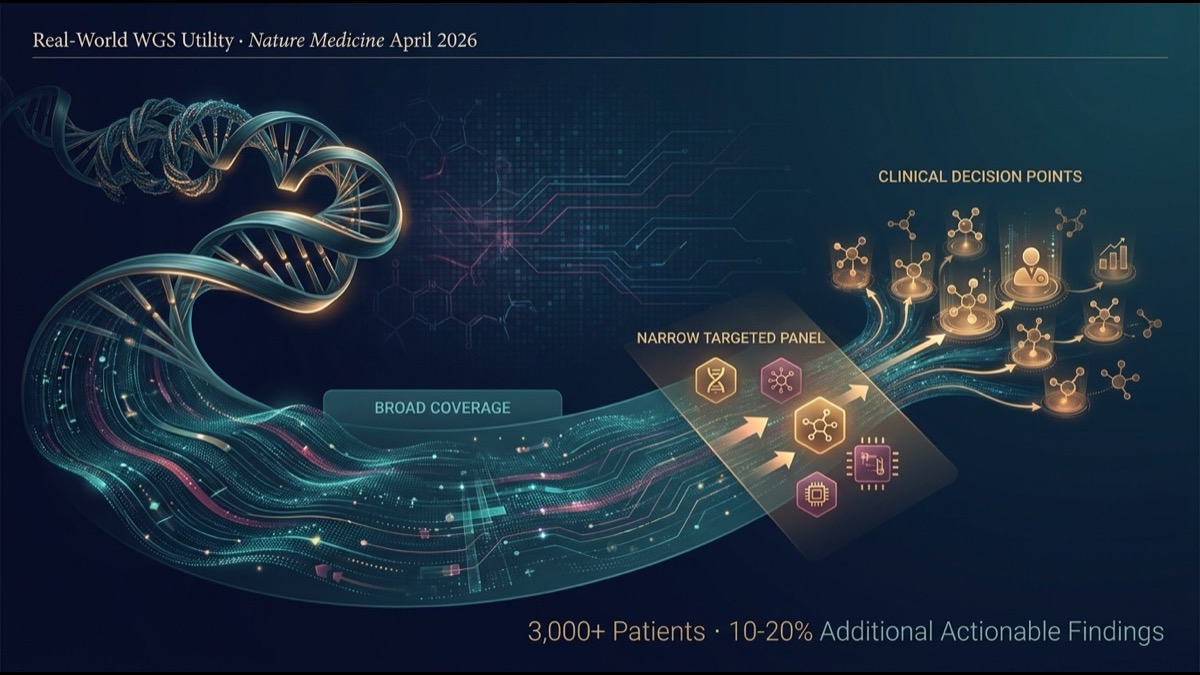
- Principal finding: WGS identifies additional actionable findings in 10-20% of patients beyond standard targeted-gene panel testing (e.g., 300-500 gene FoundationOne CDx-style panels). The advantage of WGS is most pronounced in relapsed, refractory, and rare cancer types.
- However, three operational walls remain: (1) Turnaround time (TAT) of 2-4 weeks vs. 1-2 weeks for panels, (2) Cost is 2-3× panel testing, (3) Variants of Uncertain Significance (VUS) interpretation burden on pathologists and oncologists.
- Clinical implication: “WGS for the right patient, not WGS for every patient” — a selective tiered model is the realistic answer. Centered on relapsed, refractory, rare-cancer, young-onset, and standard-treatment-failed cases, panel + WGS hierarchical combinations will become the new standard.
Introduction — “Beautiful in Research, but What About Clinic?”
Cancer treatment has undergone enormous transformation through precision oncology over the past 15 years. HER2, EGFR, ALK, KRAS, BRCA1/2, BRAF, PD-L1 — companion diagnostics targeting these molecular markers enable appropriate drug selection and extend survival.
Within this trend, targeted-gene panel testing (dozens to 500 genes) has become widely used in clinical practice. FoundationOne CDx (Foundation Medicine, 324 genes), Caris Molecular Intelligence, MSK-IMPACT (Memorial Sloan Kettering), Tempus xT — these are clinical-grade tools serving precision oncology globally.
Meanwhile, WGS (whole-genome sequencing) has accumulated massive datasets through major research programs: The Cancer Genome Atlas (TCGA), Hartwig Medical Foundation, the 100,000 Genomes Project (Genomics England UK). These have generated scientific advances in understanding cancer molecular diversity but the “individual patient’s clinical utility” has long been unclear.
The April 2026 Nature Medicine paper provides the answer at multicenter, real-world scale. This article walks through the research strategy, principal results, implementation challenges, and structural implications for the global research and industry community.
Main Body
1. Targeted Panel vs WGS — Technical Comparison
| Item | Targeted gene panel | WGS (whole-genome) |
|---|---|---|
| Coverage | 300-500 gene coding regions | Whole genome (3 billion base pairs) |
| Depth | 500-1000× (deep) | 30-100× (shallow) |
| Cost | $1,500-3,000/patient | $3,000-5,000/patient |
| TAT | 1-2 weeks | 2-4 weeks |
| Detectable variants | SNV, small indel, major fusions | SNV, indel, SV, CNV, fusion, mutational signatures all |
| Actionable findings | FDA-approved targets only | Targets + novel drivers + trial candidates |
| VUS (uncertain variants) | Few (within known scope) | Many (genome-wide discovery) |
| Report readability | Standardized | Variable, requires bioinformatics |
Targeted panels probe deep and narrow; WGS probes shallow and broad. Panels reliably detect known drug targets; WGS comprehensively detects known targets plus novel drivers, structural anomalies, and mutational signatures — the two are complementary.
2. The Paper’s Research Strategy
The team integrated real-world data from multiple major European and North American cancer centers (Hartwig Medical Foundation network, Memorial Sloan Kettering, Johns Hopkins, etc.) covering 3,000+ solid-tumor patients.
Key research questions:
- How much do panel and WGS findings overlap, and what differs?
- Did WGS-only actionable findings actually change treatment decisions and outcomes?
- How do TAT, cost, and VUS interpretation burden absorb in clinical settings?
- What is the optimal threshold for WGS deployment by cancer type and patient characteristics?
The paper answers all four with 3,000-patient real-world data.
3. Principal Result 1 — 10-20% Additional Actionable Findings
Beyond actionable findings already identified by standard panel testing, WGS identified additional actionable findings in 10-20% of patients.
Categories of additional findings:
- Rare driver mutations: SNVs/indels in genes outside panel coverage
- Large structural anomalies: chromosomal translocations, large deletions/duplications
- Mutational signatures: comprehensive determination of MMR deficiency (dMMR/MSI-H), BRCA-like / HRD (homologous recombination deficiency)
- Tumor mutational burden (TMB): more accurate calculation than panel-based
- Clonal architecture: multi-clone coexistence, evolutionary trajectory inference
HRD determination is particularly notable. While panel testing detects only major HRD-related gene mutations (BRCA1/2 etc.), WGS observes genome-wide HRD-related structural patterns, allowing PARP inhibitor indication expansion to BRCA-wild-type HRD patients.
4. Principal Result 2 — Treatment-Decision and Outcome Impact
Of patients with WGS-only additional findings:
- About half experienced treatment plan changes (clinical trial enrollment, off-label use, PARP inhibitors, etc.)
- A subset of those experienced response (complete/partial) and survival prolongation
- WGS additional findings had the largest clinical impact in standard-treatment-failed and refractory cases
This supports a positive answer: “WGS can substantively change treatment decisions.” But not for all patients — most prominently for those who have exhausted standard options.
5. Implementation Challenge 1 — TAT, Cost, VUS
Three implementation walls:
First, TAT of 2-4 weeks. About 2× the panel TAT (1-2 weeks). For progressive disease, this delays treatment initiation. Particularly disadvantageous for acute-onset cancer types (small-cell lung cancer, acute leukemia). Technical improvements (Illumina NovaSeq, PacBio Revio) are expected to compress TAT to ~1 week.
Second, $3,000-5,000/patient cost. Public-insurance reimbursement structures may not absorb this. Reimbursement architecture must evolve in parallel for WGS to scale beyond research-academic settings.
Third, VUS interpretation burden. WGS may identify thousands of VUS per patient. Clinical interpretation requires team coordination among oncologists, molecular pathologists, and bioinformatics specialists. Molecular Tumor Board infrastructure becomes essential.
6. Implementation Challenge 2 — Data Quality, Standardization, Ethics
Additional operational concerns:
Data quality and standardization. WGS results vary by institution, instrument, and sample quality. FFPE specimens degrade analysis quality compared with fresh-frozen, with high noise. Standardized pipelines (GATK, nf-core/sarek) and quality control protocols are required.
Incidental germline findings. WGS reveals germline variants (BRCA1/2, TP53, Lynch-syndrome-related, etc.). Disclosure protocols, counseling capacity, and family member testing infrastructure are needed. Patient/family psychological impact and societal implications (insurance, employment discrimination) are concerns.
Data protection and privacy. WGS data is the ultimate personal identifier. Secure storage, access control, and patient consent management require rigorous frameworks.
7. Conclusion — “WGS for the Right Patient, Not Every Patient”
The paper’s overall conclusion: WGS has clinical value in real-world settings, but “not for every patient — selective application in a tiered model” is the realistic answer.
Concrete tiered application model:
- Initial diagnosis / standard treatment initiation: standard panel testing (FoundationOne CDx etc.) screens for major actionable findings.
- Standard treatment failure / relapse: WGS, with comprehensive analysis of additional actionable findings, HRD, dMMR, TMB.
- Rare cancer / young-onset cancer / unknown primary: consider WGS at initial diagnosis.
- Integrated decision via Molecular Tumor Board: WGS results interpreted by oncology + pathology + bioinformatics team for treatment selection.
This model is hierarchical complementation, not panel-WGS substitution — optimizing cost, TAT, and interpretation burden.
8. Limitations and Caveats
First, real-world data selection bias. Participating institutions are highly WGS-motivated cancer centers; general-hospital implementation feasibility may be more limited.
Second, limited follow-up duration. How much WGS additional findings contribute to long-term survival requires 5-10 year follow-up. The paper covers 1-3 years; OS impact cannot be fully assessed.
Third, rapid technical progress. WGS TAT and cost improve at 10-15% annually, so paper-time numbers will be outdated quickly. Long-read sequencing (PacBio, Oxford Nanopore) clinical adoption will further enhance analytical capability.
Summary
- Nature Medicine April 2026 validates real-world clinical utility of solid-tumor WGS with 3,000-patient real-world data.
- WGS identifies additional actionable findings in 10-20% of patients vs standard panel, correlating with treatment plan changes, response, and survival prolongation.
- Categories: rare driver mutations, large structural anomalies, comprehensive HRD/MMR/TMB determination, clonal architecture.
- Implementation challenges: TAT 2-4 weeks, $3,000-5,000 cost, VUS interpretation burden, data standardization, incidental germline findings, privacy protection.
- Realistic answer: “WGS for the right patient, not every patient” — hierarchical complementation model centered on standard-treatment-failed, relapsed, rare cancer, young-onset cases.
- Limitations: selection bias, follow-up duration, rapid technical progress.
My Thoughts and Outlook
The paper’s significance is providing — for the long-research-biased question of WGS clinical utility — answers from large-scale real-world data. As a researcher and industry observer, I see this as the “white-boxing” milestone for precision medicine. What was a black box (“what does WGS contribute clinically?”) is now visualized with concrete numbers (10-20% additional findings, half with treatment changes, response and survival prolongation).
Three structural implications for the global ecosystem. First, WGS-clinical-grade infrastructure becomes pharma-companion-diagnostic essential. Pharma majors developing precision oncology drugs (AstraZeneca for HRD-targeting Lynparza, Merck for MSI-H Keytruda, AbbVie for KRAS-targeting RMC-9805) will increasingly require WGS-grade biomarker validation in clinical trials. Second, AI-driven VUS interpretation reshapes the bottleneck. Google DeepMind’s AlphaMissense, Tempus AI’s precision medicine platform, Foundation Medicine’s AI annotation engines are reducing the molecular-tumor-board interpretation burden. The first FDA-approved AI-VUS-classifier may emerge by 2027-28. Third, sequencing technology competition compresses cost. Illumina NovaSeq X, PacBio Revio, Oxford Nanopore PromethION — instrument competition is driving WGS toward $1,000/patient at clinical-grade quality. By 2028-30, “baseline WGS for every cancer patient” becomes a real option.
2026 is the year AI is rapidly commoditizing knowledge work. Cancer genomic interpretation sits at the intersection of “AI-augmentable” (large-scale variant interpretation, pattern recognition) and “AI-cannot-replace” (clinical decision-making, patient communication, regulatory navigation). WGS as a clinical platform plays squarely in the augmentation zone — AI will make WGS interpretation faster and cheaper, but human clinicians remain essential. The next 5-10 years will see WGS evolve from research platform to clinical standard tool.
Edited by the Morningglorysciences team.


Comments